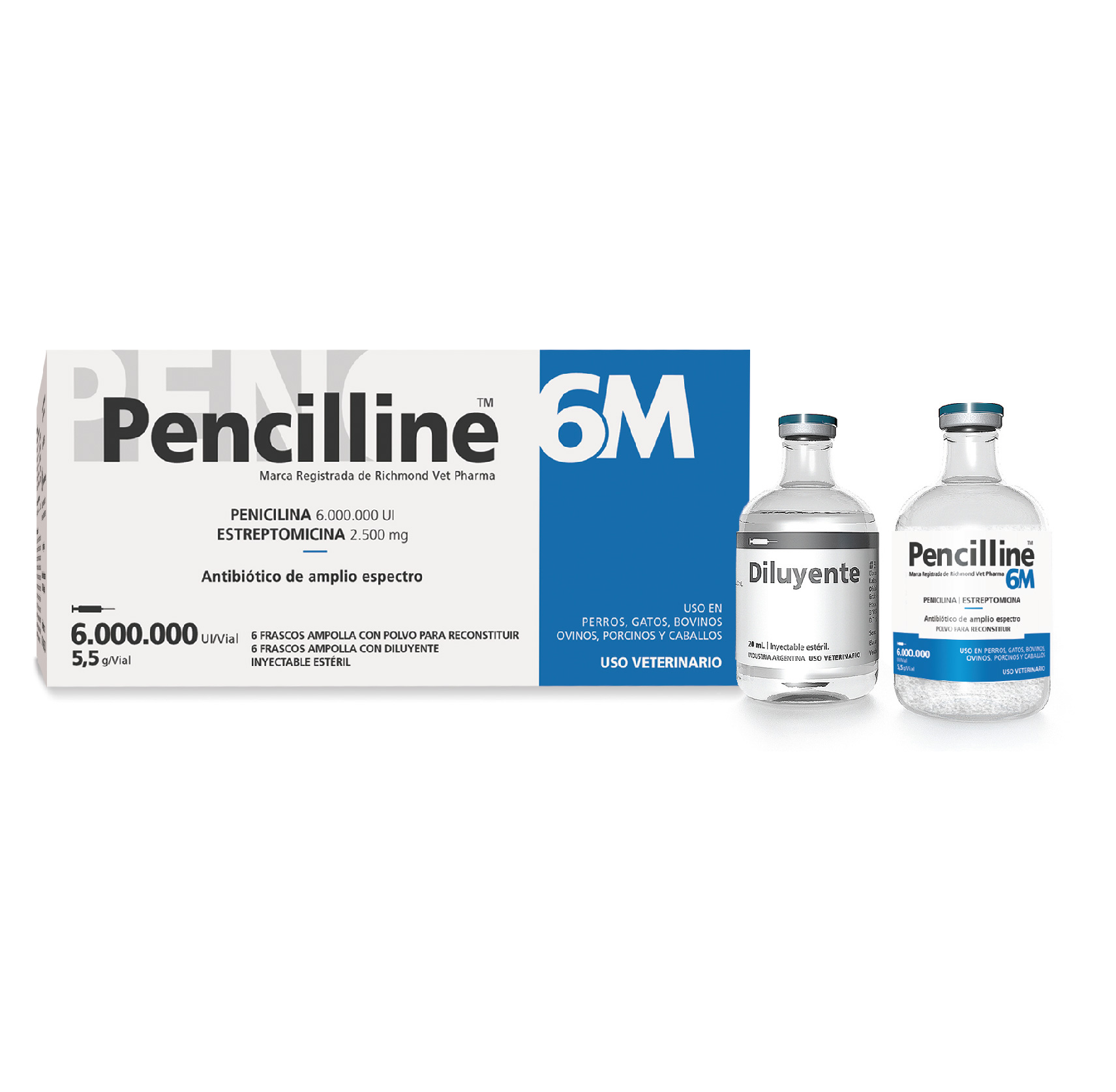
PENCILLINE 6M
Trademark of Richmond Vet Pharma S.A
VETERINARY USE
Pencilline 6M. Broad spectrum antibiotic. Penicillin 6.0 M IU & Streptomycin
Use in Dogs, Cats, Cattle, Sheep, Swine and Horses.
Composition
Each vial with powder contains:
Potassium Penicillin G 1.500.000 IU.
Procaine Penicillin G 1.500.000 IU.
Benzathine Penicillin G 3.000.000 IU.
Streptomycin Sulfate (base) 1.250 mg
Dihydrostreptomycin Sulfate (base) 1.250 mg
Each vial with diluent contains:
Water for injection q.s.f. 20 mL
Pencilline 6M, must be reconstituted for use. To do this, extract with syringe and sterile needle, 15 mL of water for injection from the vial containing the diluent and place it in the vial containing the powder to reconstitute. Shake gently until the powder completely dissolves.
After reconstitution, it is stable for 7 days if stored in refrigerator (2 to 8ºC) and 12 hours if it is kept at room temperature (15 to 30ºC). If it precipitates during storage, stir gently until the solution is reconstituted.
Indications
The combination of Penicillin and Streptomycin results in a broad spectrum bactericidal antibiotic, indicated in mixed infections caused by microorganisms sensitive to the combination of this formula. It’s indicated for abscesses, pneumonias and bronchopneumonia, bacterial or symptomatic anthrax, urinary tract infections (nephritis, pyelonephritis, and cystitis), leptospirosis, uterine infections, hoof rot, strangles in horses, tetanus, enteric toxemia, ulcerative lymphangitis, mastitis, peritonitis, neutering, tail docking, actinomycosis and actinobacillosis among others.
Administration
Deep intramuscular route.
Dosage
Minimum Indicative Dosage:
Streptomycin – Dihydrostreptomycin
Mammals: 10 mg/kg, IM Route
Penicillin
DOSAGES (IU/kg) cattle, swine, horses, dogs and cats: 8000 to 50000.
Each mL of product, reconstituted in 15 mL of diluent, contains 400.000 IU of Penicillin and 166,7 mg of Streptomycin.
Therefore it is suggested:
- Dogs and cats: 1 mL every 10 to 20 kilos of body weight.
- Sheep and swine: 1 mL every 20 to 40 kilos of body weight.
- Cattle and horses: 1 mL every 25 to 50 kilos of body weight.
It can be repeated every 3 days.
Restrictions
- Do not administer to animals with renal, hepatic and heart disease deficiencies.
- Do not administer to pregnant females.
- Do not administer to horses intended for human consumption.
- Do not administer to lactating animals.
- Do not administer to animals allergic to penicillin.
Precautions
For the reconstitution of the product, maintain the conditions of asepsis and antisepsis, respecting the storage temperature for the solution once reconstituted and the maximum duration of use after its preparation.
Shake gently before using.
The suggested dosage and warnings are in every case consideration of the acting veterinarian.
Prior to use verify that the product inviolability system and its storage conditions are adequate.
The materials used will be discarded, as will the remnants contained in the syringe. The region in which it is to be administered must be properly disinfected.
In prolonged treatments, ototoxic effects can be observed.
Store below 30 ° C, protected from light, in a dry and hygienic place.
Keep out of the reach of children.
Withdrawal period
Do not slaughter treated animals until after 30 days after the last application.
UNDER VETERINARY PRESCRIPTION ONLY
National Poison Center: 0800-333-0160
Protect the environment
SENASA (*) Cert. Nº 02-304
Produced and manufactured in facility Nº 6742:
LABORATORIOS RICHMOND DIVISIÓN VETERINARIA S.A.
Fragata Heroína 4988 | B1615ICH | Grand Bourg |Buenos Aires | Argentina
H.T.: Dr. Juan D. Onainty. MV MN 6167
Pencilline 6M
Box containing 6 vials with reconstitution powder + 6 vials with diluent.
Related products
Attention schedule:
Mon a Fri: 08:00-17:00
Where we are
Mail:
Fragata Heroína 4988 (B1615ICH)
Grand Bourg, Buenos Aires, Argentina.
Contact
Phone:
0810-333-7424
Correo electrónico:
info@richmondvet.com.ar